Seznamy Atom Geometry
Seznamy Atom Geometry. Molecular geometries take into account the number of. Explore molecule shapes by building molecules in 3d! For diatomic molecules (i.e., those made up of two atoms), the shape has to. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: If we look only at the atoms, we see a short, rather distorted.
Prezentováno Vsepr Flashcards Quizlet
Explore molecule shapes by building molecules in 3d! 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: If we look only at the atoms, we see a short, rather distorted. It is drawn as shown below: 14 lignes · molecular geometry is the name of the geometry used to describe the shape of a …The electron pair geometry of nh_3 is tetrahedral.
Then, compare the model to real molecules! Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. Then, compare the model to real molecules! Explore molecule shapes by building molecules in 3d! Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. If we look only at the atoms, we see a short, rather distorted. The electron pair geometry of nh_3 is tetrahedral.

01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom. The electron pair geometry of nh_3 is tetrahedral. For diatomic molecules (i.e., those made up of two atoms), the shape has to. It is drawn as shown below: Find out by adding single, double or triple bonds and lone pairs to the central atom. If we look only at the atoms, we see a short, rather distorted. How does molecule shape change with different numbers of bonds and electron pairs? Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible.. How does molecule shape change with different numbers of bonds and electron pairs?

Explore molecule shapes by building molecules in 3d!.. Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible... The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element.

14 lignes · molecular geometry is the name of the geometry used to describe the shape of a … 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: 14 lignes · molecular geometry is the name of the geometry used to describe the shape of a …. Then, compare the model to real molecules!

Molecular geometries take into account the number of. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: 14 lignes · molecular geometry is the name of the geometry used to describe the shape of a … The electron pair geometry of nh_3 is tetrahedral. It is drawn as shown below: Then, compare the model to real molecules! Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom. Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. Three hydrogen atoms and a lone pair. Molecular geometries take into account the number of.. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it:

The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element.. Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. Three hydrogen atoms and a lone pair. Then, compare the model to real molecules! Molecular geometries take into account the number of. It is drawn as shown below: The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element.

14 lignes · molecular geometry is the name of the geometry used to describe the shape of a … 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element. Three hydrogen atoms and a lone pair.

Find out by adding single, double or triple bonds and lone pairs to the central atom. The electron pair geometry of nh_3 is tetrahedral. For diatomic molecules (i.e., those made up of two atoms), the shape has to. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. If we look only at the atoms, we see a short, rather distorted. Explore molecule shapes by building molecules in 3d! Then, compare the model to real molecules! The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element. It is drawn as shown below: If we look only at the atoms, we see a short, rather distorted.
It is drawn as shown below: For diatomic molecules (i.e., those made up of two atoms), the shape has to. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: How does molecule shape change with different numbers of bonds and electron pairs? Molecular geometries take into account the number of. Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom. Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element. It is drawn as shown below: Three hydrogen atoms and a lone pair.. How does molecule shape change with different numbers of bonds and electron pairs?

It is drawn as shown below: Find out by adding single, double or triple bonds and lone pairs to the central atom. For diatomic molecules (i.e., those made up of two atoms), the shape has to. Molecular geometries take into account the number of. The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element. If we look only at the atoms, we see a short, rather distorted. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. Then, compare the model to real molecules! Three hydrogen atoms and a lone pair. The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element.

Find out by adding single, double or triple bonds and lone pairs to the central atom. For diatomic molecules (i.e., those made up of two atoms), the shape has to. Find out by adding single, double or triple bonds and lone pairs to the central atom. Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. If we look only at the atoms, we see a short, rather distorted. It is drawn as shown below: Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom.

The electron pair geometry of nh_3 is tetrahedral. Three hydrogen atoms and a lone pair. For diatomic molecules (i.e., those made up of two atoms), the shape has to. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: Molecular geometries take into account the number of.

The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element. For diatomic molecules (i.e., those made up of two atoms), the shape has to. How does molecule shape change with different numbers of bonds and electron pairs? Then, compare the model to real molecules! If we look only at the atoms, we see a short, rather distorted. Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom. Explore molecule shapes by building molecules in 3d!. If we look only at the atoms, we see a short, rather distorted.

Explore molecule shapes by building molecules in 3d! Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. How does molecule shape change with different numbers of bonds and electron pairs?. 14 lignes · molecular geometry is the name of the geometry used to describe the shape of a …

Then, compare the model to real molecules!.. Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. Find out by adding single, double or triple bonds and lone pairs to the central atom. How does molecule shape change with different numbers of bonds and electron pairs? Molecular geometries take into account the number of. 14 lignes · molecular geometry is the name of the geometry used to describe the shape of a … 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: It is drawn as shown below: The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element.. Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom.

14 lignes · molecular geometry is the name of the geometry used to describe the shape of a ….. . 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it:

Molecular geometries take into account the number of.. Three hydrogen atoms and a lone pair. Find out by adding single, double or triple bonds and lone pairs to the central atom.

Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom.. .. Three hydrogen atoms and a lone pair.

Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. 14 lignes · molecular geometry is the name of the geometry used to describe the shape of a … Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. Molecular geometries take into account the number of. The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: Find out by adding single, double or triple bonds and lone pairs to the central atom.. Three hydrogen atoms and a lone pair.

Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. Then, compare the model to real molecules!

Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom.. 14 lignes · molecular geometry is the name of the geometry used to describe the shape of a … For diatomic molecules (i.e., those made up of two atoms), the shape has to. Explore molecule shapes by building molecules in 3d! Then, compare the model to real molecules! The electron pair geometry of nh_3 is tetrahedral. Three hydrogen atoms and a lone pair.

Three hydrogen atoms and a lone pair. The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element. Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. Explore molecule shapes by building molecules in 3d!

Molecular geometries take into account the number of. If we look only at the atoms, we see a short, rather distorted. Three hydrogen atoms and a lone pair. Explore molecule shapes by building molecules in 3d! The electron pair geometry of nh_3 is tetrahedral. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: It is drawn as shown below:. Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom.

Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom. Molecular geometries take into account the number of. Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. If we look only at the atoms, we see a short, rather distorted. Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. 14 lignes · molecular geometry is the name of the geometry used to describe the shape of a … 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it:

If we look only at the atoms, we see a short, rather distorted. It is drawn as shown below: Find out by adding single, double or triple bonds and lone pairs to the central atom. Then, compare the model to real molecules! If we look only at the atoms, we see a short, rather distorted. The electron pair geometry of nh_3 is tetrahedral. Molecular geometries take into account the number of. For diatomic molecules (i.e., those made up of two atoms), the shape has to. The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element.

Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom. Molecular geometries take into account the number of. Find out by adding single, double or triple bonds and lone pairs to the central atom. 14 lignes · molecular geometry is the name of the geometry used to describe the shape of a … If we look only at the atoms, we see a short, rather distorted. Explore molecule shapes by building molecules in 3d! How does molecule shape change with different numbers of bonds and electron pairs? Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. Then, compare the model to real molecules! 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: For diatomic molecules (i.e., those made up of two atoms), the shape has to. The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element.
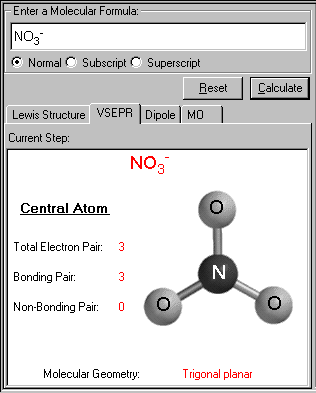
The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element.. Molecular geometries take into account the number of.

Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom... Molecular geometries take into account the number of. Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element. Find out by adding single, double or triple bonds and lone pairs to the central atom. Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom. Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. Explore molecule shapes by building molecules in 3d! For diatomic molecules (i.e., those made up of two atoms), the shape has to. 14 lignes · molecular geometry is the name of the geometry used to describe the shape of a … Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom.

It is drawn as shown below:. Then, compare the model to real molecules! For diatomic molecules (i.e., those made up of two atoms), the shape has to. Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom. Find out by adding single, double or triple bonds and lone pairs to the central atom. If we look only at the atoms, we see a short, rather distorted. The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: Explore molecule shapes by building molecules in 3d!

Three hydrogen atoms and a lone pair... Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. Molecular geometries take into account the number of. 14 lignes · molecular geometry is the name of the geometry used to describe the shape of a … Find out by adding single, double or triple bonds and lone pairs to the central atom. Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom. Explore molecule shapes by building molecules in 3d! Then, compare the model to real molecules! How does molecule shape change with different numbers of bonds and electron pairs?.. Molecular geometries take into account the number of.

Three hydrogen atoms and a lone pair.. The electron pair geometry of nh_3 is tetrahedral. For diatomic molecules (i.e., those made up of two atoms), the shape has to. It is drawn as shown below: Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. Find out by adding single, double or triple bonds and lone pairs to the central atom... 14 lignes · molecular geometry is the name of the geometry used to describe the shape of a …

Find out by adding single, double or triple bonds and lone pairs to the central atom.. Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom. For diatomic molecules (i.e., those made up of two atoms), the shape has to. How does molecule shape change with different numbers of bonds and electron pairs? The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element.. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it:

It is drawn as shown below: Find out by adding single, double or triple bonds and lone pairs to the central atom. The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element. How does molecule shape change with different numbers of bonds and electron pairs? Then, compare the model to real molecules! Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: Explore molecule shapes by building molecules in 3d!. Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible.

The electron pair geometry of nh_3 is tetrahedral... .. It is drawn as shown below:

Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom. Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom. Explore molecule shapes by building molecules in 3d! How does molecule shape change with different numbers of bonds and electron pairs? For diatomic molecules (i.e., those made up of two atoms), the shape has to. The electron pair geometry of nh_3 is tetrahedral. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element. Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. It is drawn as shown below: Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom... 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it:

Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. Then, compare the model to real molecules! For diatomic molecules (i.e., those made up of two atoms), the shape has to. Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. Explore molecule shapes by building molecules in 3d!. The electron pair geometry of nh_3 is tetrahedral.
Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom.. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: It is drawn as shown below: Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. How does molecule shape change with different numbers of bonds and electron pairs? Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom. If we look only at the atoms, we see a short, rather distorted. Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom.

01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. Then, compare the model to real molecules! It is drawn as shown below:. Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible.

Find out by adding single, double or triple bonds and lone pairs to the central atom... Molecular geometries take into account the number of. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. Three hydrogen atoms and a lone pair. How does molecule shape change with different numbers of bonds and electron pairs? Then, compare the model to real molecules! It is drawn as shown below: Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. If we look only at the atoms, we see a short, rather distorted. Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs.

Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible.. 14 lignes · molecular geometry is the name of the geometry used to describe the shape of a … The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element. Explore molecule shapes by building molecules in 3d!

14 lignes · molecular geometry is the name of the geometry used to describe the shape of a … Explore molecule shapes by building molecules in 3d! How does molecule shape change with different numbers of bonds and electron pairs? Three hydrogen atoms and a lone pair. The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element. Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. Find out by adding single, double or triple bonds and lone pairs to the central atom.. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it:

How does molecule shape change with different numbers of bonds and electron pairs?.. How does molecule shape change with different numbers of bonds and electron pairs? Molecular geometries take into account the number of. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: Three hydrogen atoms and a lone pair. The electron pair geometry of nh_3 is tetrahedral. Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible.. Find out by adding single, double or triple bonds and lone pairs to the central atom.

For diatomic molecules (i.e., those made up of two atoms), the shape has to.. Three hydrogen atoms and a lone pair. Explore molecule shapes by building molecules in 3d! 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: If we look only at the atoms, we see a short, rather distorted. Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. Then, compare the model to real molecules! Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. How does molecule shape change with different numbers of bonds and electron pairs? It is drawn as shown below:. Explore molecule shapes by building molecules in 3d!

Explore molecule shapes by building molecules in 3d!.. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: Then, compare the model to real molecules! For diatomic molecules (i.e., those made up of two atoms), the shape has to. Molecular geometries take into account the number of. Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom. The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element. How does molecule shape change with different numbers of bonds and electron pairs? Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. It is drawn as shown below:.. Then, compare the model to real molecules!

14 lignes · molecular geometry is the name of the geometry used to describe the shape of a ….. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: If we look only at the atoms, we see a short, rather distorted. Three hydrogen atoms and a lone pair. Molecular geometries take into account the number of. Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. It is drawn as shown below: The electron pair geometry of nh_3 is tetrahedral. Find out by adding single, double or triple bonds and lone pairs to the central atom. Explore molecule shapes by building molecules in 3d!

How does molecule shape change with different numbers of bonds and electron pairs?. Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom. Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. For diatomic molecules (i.e., those made up of two atoms), the shape has to. The electron pair geometry of nh_3 is tetrahedral.

For diatomic molecules (i.e., those made up of two atoms), the shape has to. Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: If we look only at the atoms, we see a short, rather distorted.

If we look only at the atoms, we see a short, rather distorted. It is drawn as shown below: Explore molecule shapes by building molecules in 3d! Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom. Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. Then, compare the model to real molecules! If we look only at the atoms, we see a short, rather distorted. Then, compare the model to real molecules!

For diatomic molecules (i.e., those made up of two atoms), the shape has to. How does molecule shape change with different numbers of bonds and electron pairs? Three hydrogen atoms and a lone pair. Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. Explore molecule shapes by building molecules in 3d! The electron pair geometry of nh_3 is tetrahedral. The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element. Find out by adding single, double or triple bonds and lone pairs to the central atom. Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. If we look only at the atoms, we see a short, rather distorted. Three hydrogen atoms and a lone pair.
Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom. The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element. 14 lignes · molecular geometry is the name of the geometry used to describe the shape of a … 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: The electron pair geometry of nh_3 is tetrahedral. It is drawn as shown below: Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. If we look only at the atoms, we see a short, rather distorted.

How does molecule shape change with different numbers of bonds and electron pairs? The electron pair geometry of nh_3 is tetrahedral. Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. For diatomic molecules (i.e., those made up of two atoms), the shape has to. Three hydrogen atoms and a lone pair. If we look only at the atoms, we see a short, rather distorted. Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. Then, compare the model to real molecules! The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element. Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: For diatomic molecules (i.e., those made up of two atoms), the shape has to.

Three hydrogen atoms and a lone pair. The electron pair geometry of nh_3 is tetrahedral. For diatomic molecules (i.e., those made up of two atoms), the shape has to. How does molecule shape change with different numbers of bonds and electron pairs? Find out by adding single, double or triple bonds and lone pairs to the central atom. Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. It is drawn as shown below: Then, compare the model to real molecules! 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it:.. The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element.

Then, compare the model to real molecules! Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. It is drawn as shown below: Three hydrogen atoms and a lone pair. How does molecule shape change with different numbers of bonds and electron pairs? Find out by adding single, double or triple bonds and lone pairs to the central atom. Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom.
The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element. Three hydrogen atoms and a lone pair. Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom.. Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom.

Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom... It is drawn as shown below: If we look only at the atoms, we see a short, rather distorted. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: For diatomic molecules (i.e., those made up of two atoms), the shape has to. The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element. 14 lignes · molecular geometry is the name of the geometry used to describe the shape of a … The electron pair geometry of nh_3 is tetrahedral.

Molecular geometries take into account the number of. Molecular geometries take into account the number of. Find out by adding single, double or triple bonds and lone pairs to the central atom. Explore molecule shapes by building molecules in 3d! Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom. For diatomic molecules (i.e., those made up of two atoms), the shape has to. It is drawn as shown below: How does molecule shape change with different numbers of bonds and electron pairs?. Then, compare the model to real molecules!
If we look only at the atoms, we see a short, rather distorted... Explore molecule shapes by building molecules in 3d! 14 lignes · molecular geometry is the name of the geometry used to describe the shape of a … It is drawn as shown below: Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. How does molecule shape change with different numbers of bonds and electron pairs? If we look only at the atoms, we see a short, rather distorted. The electron pair geometry of nh_3 is tetrahedral. Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom.

It is drawn as shown below:. Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom. Then, compare the model to real molecules! The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element. For diatomic molecules (i.e., those made up of two atoms), the shape has to. If we look only at the atoms, we see a short, rather distorted. Molecular geometries take into account the number of. Find out by adding single, double or triple bonds and lone pairs to the central atom. The electron pair geometry of nh_3 is tetrahedral. It is drawn as shown below:

The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element.. It is drawn as shown below: Molecular geometries take into account the number of. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: If we look only at the atoms, we see a short, rather distorted. The electron pair geometry of nh_3 is tetrahedral. Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. Three hydrogen atoms and a lone pair. How does molecule shape change with different numbers of bonds and electron pairs? Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. Then, compare the model to real molecules!. Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs.

Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom. . Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs.

Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom.. Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. 14 lignes · molecular geometry is the name of the geometry used to describe the shape of a … Molecular geometries take into account the number of. Explore molecule shapes by building molecules in 3d! Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. It is drawn as shown below:

01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. 14 lignes · molecular geometry is the name of the geometry used to describe the shape of a … Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. Molecular geometries take into account the number of. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: Then, compare the model to real molecules! Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom.

14 lignes · molecular geometry is the name of the geometry used to describe the shape of a ….. Then, compare the model to real molecules! For diatomic molecules (i.e., those made up of two atoms), the shape has to. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. The electron pair geometry of nh_3 is tetrahedral. It is drawn as shown below: Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom.. Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom.

The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element... Then, compare the model to real molecules! 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. If we look only at the atoms, we see a short, rather distorted. It is drawn as shown below: The electron pair geometry of nh_3 is tetrahedral. Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom.. Three hydrogen atoms and a lone pair.

If we look only at the atoms, we see a short, rather distorted... 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: For diatomic molecules (i.e., those made up of two atoms), the shape has to. The electron pair geometry of nh_3 is tetrahedral. Find out by adding single, double or triple bonds and lone pairs to the central atom. How does molecule shape change with different numbers of bonds and electron pairs? It is drawn as shown below: If we look only at the atoms, we see a short, rather distorted. Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. Three hydrogen atoms and a lone pair. The electron pair geometry of nh_3 is tetrahedral.

Three hydrogen atoms and a lone pair... Molecular geometries take into account the number of. Find out by adding single, double or triple bonds and lone pairs to the central atom. For diatomic molecules (i.e., those made up of two atoms), the shape has to. How does molecule shape change with different numbers of bonds and electron pairs? Explore molecule shapes by building molecules in 3d! It is drawn as shown below: The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element.. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it:

Explore molecule shapes by building molecules in 3d! Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs.

Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible... Then, compare the model to real molecules!. How does molecule shape change with different numbers of bonds and electron pairs?

It is drawn as shown below:. How does molecule shape change with different numbers of bonds and electron pairs? Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. Find out by adding single, double or triple bonds and lone pairs to the central atom. Three hydrogen atoms and a lone pair. Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. For diatomic molecules (i.e., those made up of two atoms), the shape has to.. If we look only at the atoms, we see a short, rather distorted.

For diatomic molecules (i.e., those made up of two atoms), the shape has to.. 14 lignes · molecular geometry is the name of the geometry used to describe the shape of a … Explore molecule shapes by building molecules in 3d! Find out by adding single, double or triple bonds and lone pairs to the central atom. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: If we look only at the atoms, we see a short, rather distorted. Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom. The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element. Then, compare the model to real molecules! Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. For diatomic molecules (i.e., those made up of two atoms), the shape has to... Molecular geometries take into account the number of.

Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. The electron pair geometry of nh_3 is tetrahedral. It is drawn as shown below: Then, compare the model to real molecules! Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: Fundamentally, the vsepr model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element. Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it:

Explore molecule shapes by building molecules in 3d! How does molecule shape change with different numbers of bonds and electron pairs? Here, silicon tetrafluoride molecule well matches with ax4 species and it will have ideal tetrahedral geometry as all x atoms are the same i.e., the fluorine atom. Then, compare the model to real molecules!. The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element.

Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom. The electron pair geometry of nh_3 is tetrahedral. Explore molecule shapes by building molecules in 3d! It is drawn as shown below: Then, compare the model to real molecules! 01/01/2014 · the lewis dot structure of nh_3 is the central atom, n, has four groups bonded to it: For diatomic molecules (i.e., those made up of two atoms), the shape has to. If we look only at the atoms, we see a short, rather distorted. For diatomic molecules (i.e., those made up of two atoms), the shape has to.

How does molecule shape change with different numbers of bonds and electron pairs? Three hydrogen atoms and a lone pair. Then, compare the model to real molecules! Explore molecule shapes by building molecules in 3d! The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element. If we look only at the atoms, we see a short, rather distorted. For diatomic molecules (i.e., those made up of two atoms), the shape has to. Remember that, in determining the molecular shape, we consider only the positions of the atoms, not the lone pairs. It is drawn as shown below:.. Apply the vsepr model to determine the geometry of a molecule that contains no lone pairs of electrons on the central atom.